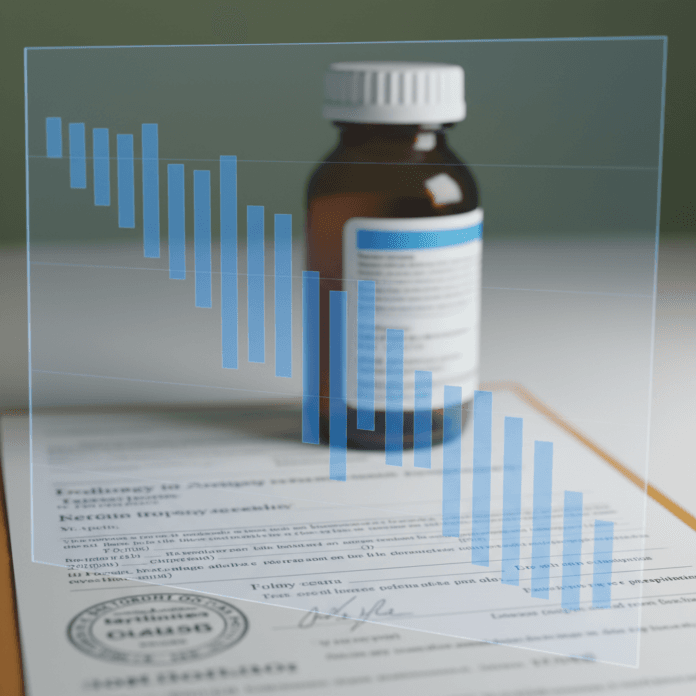
Australia’s medical cannabis sales experienced a 28.5% decline in the second half of 2025 compared to the first half, marking the first significant reduction since the national program’s establishment in 2016. This downturn in medical cannabis sales is detailed in a new report from the Penington Institute, an independent Australian not-for-profit focused on drug-related harm reduction. The Institute attributes this decline primarily to increased regulatory enforcement targeting high-volume medical clinics and prescribers, as reported by GN: medicinal cannabis.
The Penington Institute’s report indicates that medical cannabis sales in Australia peaked at 3.72 million units in the second half of 2024 before falling to 2.65 million units in the second half of 2025.
Regulatory Scrutiny and Enforcement Actions
The decline in medical cannabis sales follows a period of heightened scrutiny from key industry bodies and regulators. In 2025, the Australian Medical Association (AMA) and the Pharmacy Guild of Australia jointly wrote to the nation’s Health Minister. Their letter urged the Australian government to address the largely unregulated increase in medicinal cannabis prescribing and dispensing. Key recommendations included:
- Strengthening clinical governance through national safety and quality bodies.
- Supporting the Australian Health Practitioner Regulation Agency (Ahpra) in regulating prescribing and dispensing practices.
- Reforming the Special Access Scheme to restrict its use to exceptional cases under supervision.
- Encouraging sponsors to register products on the Australian Register of Therapeutic Goods (ARTG) for evidence-based indications within two years.
The framework allowing Australians to access medical cannabis via a telehealth model came under particular examination. This followed a rapid acceleration in sales from 2023, driven by high-volume clinics that, according to the Penington Institute, sometimes prioritized commercial interests over patient care through practices such as brief consultations, unlawful advertising, and “closed loop” business models.
Regulator Interventions
In response to these concerns, Ahpra published new clinical guidance in July 2025, reminding healthcare practitioners to prioritize patient wellbeing. By mid-2025, Ahpra had taken enforcement action against more than 50 medical practitioners. Concurrently, Australia’s Therapeutic Goods Administration (TGA) issued dozens of infringement notices to six cannabis businesses and two individuals for alleged unlawful advertising of medicinal cannabis on their websites and social media platforms.
In September, the TGA announced it had issued six infringement notices totaling $118,800 and a direction notice to Dispensed Pty Ltd (Dispensed) for alleged unlawful advertising of medicinal cannabis, contravening Australian regulations.
Market Dynamics and Patient Access Considerations
While regulatory enforcement has been a factor in the recent trend of medical cannabis sales, the Penington Institute emphasizes the importance of balancing enforcement with protecting patient access. Recent research from the University of Sydney’s Lambert Initiative found that most Australians continue to source cannabis from illicit channels, despite an increase in access to prescription products.
The Penington Institute concluded that “Protecting Australian medicinal patients requires regulators to enforce appropriate rules effectively.” The report further noted that “commercially driven structures and associated high-volume prescribing have been the weakest points in safeguarding patient wellbeing” and that “the recent increase in regulatory compliance and enforcement represents a positive development and is likely a key factor in the observed decline in medicinal cannabis sales in late 2025.”
Overview of the Australian Medical Cannabis Market
As of December 31, 2025, Australia’s Therapeutic Goods Administration (TGA) had listed 877 cannabis products. These products are categorised into five types:
- CBD medicinal cannabis products (CBD ≥98%)
- CBD-dominant medicinal cannabis products (CBD ≥60% and <98%)
- Balanced medicinal cannabis products (CBD ≥40% and <60%)
- THC or other cannabinoid dominant medicinal cannabis products (CBD ≥2% and <40%)
- THC or other cannabinoid medicinal cannabis products (CBD <2%)
Data from Australia’s Office of Drug Control (ODC) indicates that the total quantity of cannabis imported into Australia under the country’s cannabis for medical purposes regulations increased from nearly 45,000 kilograms in 2023 to more than 77,000 kilograms in 2024. Canada remains the largest exporter, accounting for 80% of Australia’s total import volume in 2024, with Canadian imports rising 73% from 35,941 kg in 2023 to 62,111 kg in 2024.
International Regulatory Context
Australia is not isolated in its efforts to manage the medical cannabis sector. Draft legislation in Germany’s Cabinet seeks to amend the country’s Medical Cannabis Act to prohibit online or telemedicine access. Similarly, in Canada, the health regulator has been addressing concerns regarding physicians linked to a high number of high-gram-per-day cannabis authorizations, often associated with large personal or designated production licenses.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Hemp Gazette does not provide medical recommendations, diagnoses, or treatment plans. Always consult a qualified healthcare practitioner before making any decisions regarding your health or any medical condition. Statements concerning the therapeutic uses of hemp, cannabis, or cannabinoid-derived products have not been evaluated by Australia’s Therapeutic Goods Administration (TGA). Medicinal cannabis products in Australia are accessed via prescription pathways under TGA regulation.